|
group 1A: 1+ group 6A: 2-group 5A: 3-group 2A: 2+ In what group of the periodic table would elements that form ions with the following charges likely be found: 2- 3+ 1+ 2+ Group 6A Group 3A Group 1A Group 2A. An iron ion with a charge of +8 is not very likely, therefore, the octet rule is not applicable to transition elements. What are the charges on simple ions formed from atoms of group 1A elements 6A elements 5A elements 2A elements. In order for an element such as iron (Fe) to achieve the same noble gas configuration of argon (Ar), it would need to lose 6 electrons in the 3 d subshell and 2 electrons in the 4s subshell. The image below shows the general trend for ionic charge. But you need to know the specific ionic charge elments. Answer c Because this element is located in Group 18, or 8A, on the periodic table, it is a noble gas and, therefore, will not ionize. The suffix of this element's name is replaced with '-ide' to indicate the negative charge of the anion that it forms. Non-metals (found on the right) will be negative. The resultant ion is symbolized as P-3 and is named the phosphide ion. 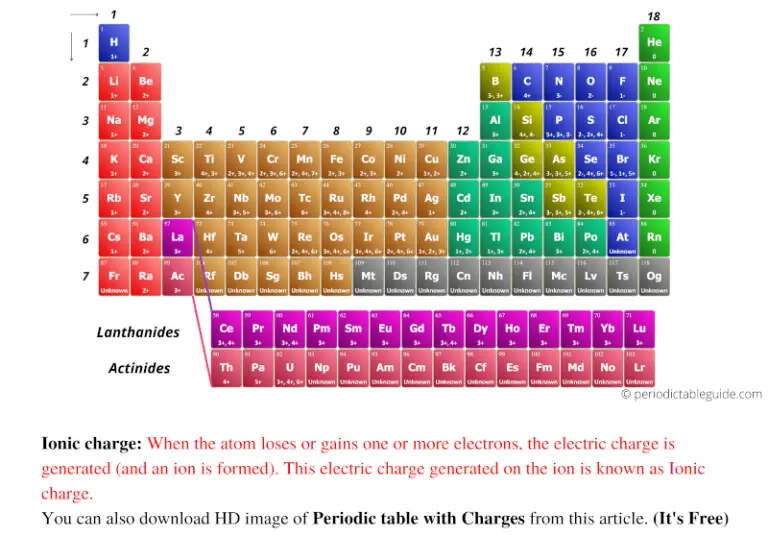
On the Periodic Table metals (found on the left of the table) will be positive. This is because the transition metals have electrons in d subshell and do not follow the octet rule. To find the ionic charge of an element youll need to consult your Periodic Table. Notice that there is no simple pattern for transition metal ions (or for the larger main group elements) as there is with the main group ions.  
\) shows the characteristic charges for some of these ions.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
